ABSTRACT
Peripheral neuropathy is one of the most common neurological problems encountered by physicians, and the broad diagnosis of neuropathy has a wide variety of underlying etiologies and varied clinical presentations. This review aims to cover the spectrum of treatable neuropathies, their etiologies, diagnostic criteria, and advances in treatment. Clinical pattern recognition and neuropathy characterization help narrow the list of differential diagnoses and thus direct the investigations. The list of treatable neuropathies is increasing and mainly includes metabolic, immune, infectious, toxic, and nutritional etiologies. Prompt recognition of treatable neuropathies with early treatment reduces morbidity and disability. In this review, we shall discuss the common treatable neuropathies in detail and tabulate the uncommon conditions.
How to cite this article
Khadilkar S, Mahajan J, Halani H, et al. Treatable Neuropathies. J Assoc Physicians India 2025;73(9):73-84.
INTRODUCTION
Peripheral neuropathy is a spectrum of diseases affecting a peripheral nerve from preganglionic roots, roots, dorsal root ganglion (DRG), postganglionic roots, plexus, and trunk to single or multiple nerves. Neuropathic disorders can affect the cell body (neuronopathy) or they can affect peripheral processes (peripheral neuropathy). 1 Neuronopathies mainly affect the DRG and anterior horn cells, named sensory ganglionopathy or ganglionopathy and motor neuron diseases. Peripheral neuropathies can be classified as per their etiologies, types of nerve fibers affected (sensory, motor, or autonomic), and the portion of the nerve fiber affected (axon or myelin). A proportion of neuropathies respond to treatment. Treatable neuropathies have the potential for treatment and hence reversibility or remission, as seen in chronic inflammatory demyelinating polyneuropathy (CIDP) and its variants, drug/toxin-induced neuropathies, and infectious neuropathies. The groups of treatable neuropathies are many, and some of these conditions are more common than others in clinical practice. Table 1 gives a comprehensive list of the treatable neuropathies with their salient clinical and investigative features and treatments. The common treatable neuropathies are discussed in some detail in this review.
List of treatable neuropathies with salient features and treatment
| Etiology | Clinical hallmarks | Treatment strategies |
|---|---|---|
| Metabolic | ||
| DM | Distal symmetric axonopathy usually with a chronic course, fluctuations in relation to glycemic control Small fiber predominant neuropathy early in the course Less often, asymmetric, painful affection of roots/plexus/isolated nerves | Adequate glycemic control and correction of other metabolic factors like hyperlipidemia, obesity, etc. Drugs for neuropathic pain Trial of immunotherapy in proper clinical settings |
| Hypothyroidism | Carpel tunnel syndrome Distal symmetric sensorimotor polyneuropathy 2 | Thyroid hormone supplements CTS may require decompression additionally 3 |
| Renal failure | Distal axonopathy 4 | Correction of uremia, for example, hemodialysis, transplantation 5 |
| Critical illness polyneuropathy | Generalized sensorimotor polyneuropathy with or without myopathy 6 | Aggressive neurorehabilitation, avoidance of steroids, neuromuscular blockers, and control of hyperglycemia and infections |
| Celiac disease | Generalized sensorimotor polyneuropathy, small fiber neuropathy, and neuromyotonia | Gluten-free diet may prevent neuropathy and halt the progress of neuropathy 7 |
| Immunological | ||
| AIDP | Acute onset nonlength dependent polyradiculoneuropathy with additional facial, bulbar, respiratory, or autonomic involvement and a preceding history of infection (refer to text for the common and uncommon variants) | IVIG, PE, and complement inhibitors |
| CIDP | Subacute or chronic or relapsing-remitting course of nonlength dependent sensorimotor weakness and areflexia usually in a middle-aged male Atypical presentations are known as pure sensory or motor involvement, distal predominant or multifocal involvement, pure sensory ataxia, etc. (refer to the text for details) | Steroids, IVIG, PE, Rtx Maintenance immunosuppressant drugs—AZA and MMF |
| Vasculitis | Acute/subacute onset mononeuritis multiplex, painful, asymmetric, or multifocal involvement Look for systemic signs of rheumatological disorders, vasculitic skin lesions 8 | Immunomodulatory drugs (Mtx, AZA, MMF, cyclophosphamide, IVIG, etc.) |
| Infections | ||
| Leprosy | Commonly, mononeuritis multiplex with predominant loss of pain and temperature sensations Look for hypopigmented and hypoesthetic skin patches and thickened nerves | MDT as per WHO recommendations |
| HIV | Distal symmetric axonopathy is common in chronic infection or related to HAART AIDP and CIDP both presentations are known to occur | HAART, supportive management for neuropathic pain |
| Cytomegalovirus (CMV) | Lumbosacral polyradiculopathy Mononeuritis multiplex 9 | Ganciclovir, foscarnet, and cidofovir as monotherapy |
| Lyme's disease | Bilateral facial neuropathy most common Asymmetric polyradiculoneuropathy Mononeuritis multiplex Primary axonopathy Associated systemic features like rash, fever | Cephalosporins and amoxycillin |
| Toxic | ||
| Drugs like antibiotics, chemotherapy | Detailed account of all the drugs/alternative medicines and temporal relation to the neuropathic symptoms should be noted Sensory involvement is more common, whereas motor predominance can be seen in lead toxicity, GBS-like syndrome with arsenic or thallium 10 | Removal of the toxin Chelation therapy in metal exposure Supportive measures |
| Heavy metals like lead, arsenic, thallium, mercury etc. | ||
| Alcohol related | ||
| Nutritional | ||
| Vit B12 deficiency | Sensorimotor axonopathy involving large fibers—loss of kinesthetic sense Additional visual or cognitive changes Myelopathy in the form of SACD Systemic signs, for example, skin pigmentation, clinical settings for malabsorption etc. | Vitamin B12 and folate supplements |
| Other causes are B6 deficiency, thiamine deficiency, and Vit E deficiency | Large fiber sensory predominant axonopathy Systemic signs: atrophic skin changes, cognitive impairment, background of malnutrition, or alcoholism (thiamine deficiency) Disorders of fat absorption and lipoproteins, additional spinocerebellar involvement (Vit E deficiency) | Nutritional supplements |
Commonly encountered treatable neuropathies are discussed below under the following headings:
Metabolic-related neuropathy—diabetic and other uncommon neuropathies.
Immune-mediated neuropathies—acute inflammatory demyelinating polyneuropathy (AIDP), CIDP, and others.
Infections-related neuropathy—leprous neuropathy and human immunodeficiency virus (HIV)-related neuropathy.
Drug/toxin induced neuropathy.
Cranial neuropathies.
Entrapment neuropathies.
Autonomic neuropathy.
METABOLIC-RELATED NEUROPATHY
Diabetes-related Peripheral Neuropathy
Diabetic neuropathy (DN) is the most common cause of peripheral neuropathy worldwide. As per the Diabetes Control and Complications Trial (DCCT), the prevalence of DN is approaching almost 50% in both type 1 and type 2 diabetes mellitus (DM). 11 DN has a much higher incidence in type 2 DM (6,100 per 1,00,000 person-years) compared to type 1 DM (2,800 per 1,00,000 person-years). 12
Risk Factors
The duration of diabetes and glycosylated/glycated hemoglobin (HbA1c) levels are among the most important risk factors for DN and other microvascular complications. 13 Other metabolic factors that are most associated with diabetes, such as hypertension, hypertriglyceridemia, abdominal obesity, uric acid levels, and low high-density lipoprotein (HDL), are also major predictors of DN. Metabolic syndrome, along with its components, has been found to be an independent risk factor for peripheral neuropathy, especially sensory peripheral neuropathy. 14 Prediabetes is the earliest stage of glucose dysregulation, including the impaired glucose tolerance (IGT) test or impaired fasting glucose (IFG) (American Diabetes Association guidelines). The recent MONICA/KORA trial demonstrated that neuropathy was more common in IGT as compared to the control group, with preferential small fiber involvement in the prediabetes group. 15
Clinical Manifestations
Diabetic neuropathy involves varied neurological patterns of neuropathy. Distal symmetric polyneuropathy (DSPN) is the most common pattern encountered in DM, which can affect predominantly small fibers, large fibers, or both together. Following is the spectrum of DN, starting from roots on the left to the peripheral nerve on the right ( Table 2 and Fig. 1 ).
Spectrum of DN
| Radiculoplexopathy | Type C nerve fibers/autonomic neuropathy | Mononeuropathy | DSPN |
|---|---|---|---|
|
|
|
|
Spectrum of DN affecting different levels of a peripheral nerve
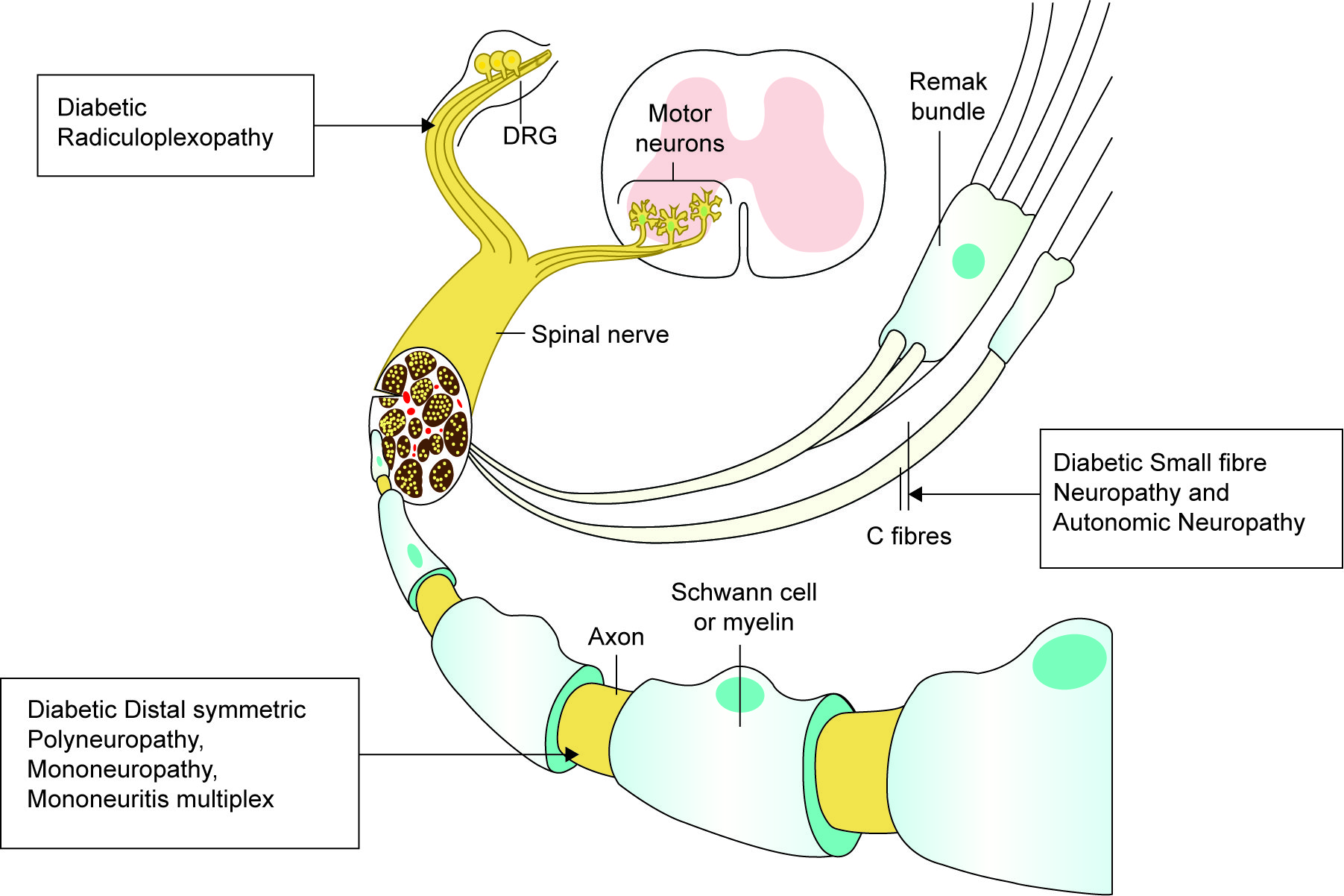
Management of Diabetic Neuropathy
Prevention of Diabetic Neuropathy
Periodic assessments for neuropathy with clinical sensory testing with 10 gm monofilament test, 16 correcting any coexistent B12 deficiency, 17 optimizing glycemic control, 10 , 16 a high suspicion for autonomic involvement, and foot care is important in the prevention, early detection, and reducing the complications of DN.
Pharmacological and Nonpharmacological Treatment of Diabetic Neuropathy
The stability of HbA1c levels is more important than the actual level of control in the treatment of DN. Long-term follow-up of the landmark DCCT demonstrated that more intensive glucose control ameliorated the onset of neuropathy as well as the progression of surrogate electrophysiologic markers of neuropathy. 11 , 21 The Food and Drug Administration (FDA)-approved first-line therapy includes duloxetine, pregabalin, gabapentin, venlafaxine, and amitriptyline. 22 - 24 The time to peak response is different for different drugs, but in general, 2–3 months is typically required for titration and to gauge the initial response of medications. In patients with inadequate response to initial treatment, either switching to second-line therapy or the addition of second first-line therapy is recommended. Topical treatments with capsaicin 8% with or without lignocaine are effective local therapy for DN-related pain. 24 , 25 Nutraceutical agents like alpha-lipoic acid (ALA), benfotiamine, acetyl-L-carnitine, gamma-linolenic acid (GLA), vitamin B12, and vitamin D3 are a few of the important molecules that have shown modest decreases in pain related to DN. Table 3 depicts the medications that can be used in the management of pain.
Medication to treat neuropathic pain
| Drug class | Dose | Comorbidities favoring use | Comorbidities favoring avoidance | Side-effects |
|---|---|---|---|---|
| Serotonin norepinephrine reuptake inhibitors | ||||
| Duloxetine—starting dose of 20–30 mg/day titrated up to a max of 60–120 mg/day | Depression and anxiety | Restless leg syndrome Sexual dysfunction Angle-closure glaucoma 22 | Nausea, somnolence, dizziness, decreased appetite, constipation, diaphoresis, and sexual dysfunction 22 , 24 |
| Tricyclic antidepressants (TCA) | ||||
| Amitriptyline | Starting dose—10–25 mg/day titrated up to a maximum 200 mg/day | Depression, anxiety, and insomnia | Dry mouth, somnolence, and urinary retention | Cardiac dysfunction, prolonged Qtc, and orthostatic hypotension |
| Gabapentoid antiseizure medications | ||||
| Gabapentin, pregabalin | Starting dose of 75–150 mg/day titrated up to a maximum dose of 600 mg/day | Restless leg syndrome, essential tremor, and insomnia | COPD and substance abuse | Peripheral edema, weight gain, somnolence, and dizziness 23 |
IMMUNE-MEDIATED NEUROPATHIES
Acute Inflammatory Demyelinating Polyneuropathy
Acute inflammatory demyelinating polyneuropathy or Guillain–Barré syndrome (GBS) is acute immune-mediated polyneuropathy presenting as a variable degree of symmetrical ascending weakness of all limbs with occasional respiratory failure and autonomic dysfunction that reaches maximal severity within 4 weeks. The main pathophysiology behind GBS is molecular mimicry between microbial and nerve antigens causing an aberrant autoimmune response. Preceding infection, mostly by Campylobacter jejuni , triggers an autoimmune response that mainly targets peripheral nerves and their spinal roots. The variants of GBS include:
| 1. Acute inflammatory demyelinating polyneuropathy | |
| IgG autoantibodies to GM1 and GD1a |
| |
| IgG autoantibodies to GT1a, GQ1b, and GD1a |
| |
| 2. Miller Fischer syndrome | |
| IgG autoantibodies to GM1 and GD1a |
| |
| 3. CNS variant—Bickerstaff's Brainstem encephalitis | |
Given the immune-mediated nature of the illness, standard treatment includes intravenous immunoglobulin (IVIG) and plasma exchange (PE). One or the other should be started as soon as possible after the diagnosis of GBS has been made. PE was reported to be first used in GBS between 1978 and 1981. PE is a therapeutic procedure that separates plasma from cells using a filter in a dialysis machine, where cells are reinfused back into circulation and plasma is removed and replaced with either fresh frozen plasma (FFP) or reconstituted human protein (albumin). Trials comparing the efficacy of IVIG with PE showed equal efficacy in decreasing hospital stay, hastening recovery, and preventing mechanical ventilation and respiratory depression. 26 - 28 Different dosages of IVIG (0.4 gm/kg/day) were administered over 3 vs 6 days, which showed the time required to regain the ability to walk with assistance was shorter in the latter group with lower infusion-related side effects. 29 For a proportion of patients with refractory GBS, the utility of a second course of IVIG was studied in a recent RCT (SID-GBS), but the outcomes were negative. 30
Treatment-related fluctuations (TRF) are often seen in 10% of GBS within 2 weeks to 2 months after first treatment initiation. 31 Retrospective studies have shown that treating with more than one modality in cases of clinical deterioration, lack of improvement, or TRF has no significant benefits. Distinguishing such cases from acute-onset CIDP or subacute inflammatory demyelinating polyneuropathy (SIDP) becomes important since the therapeutic and prognostic implications vary. Recently, a new method has been experimented with in children, known as the “Zipper method,” using immediate IVIG after each session of PE in nine patients, which has been shown to reduce mortality, 32 , 33 speed up weaning from mechanical ventilation, and shorten hospital stay, although RCT is not available for this method and hence the efficiency of this method is currently questionable. Eculizumab, a humanized monoclonal antibody against the complement protein C5, was tested in two randomized, double-blind, placebo-controlled phase 2 trials. Neither showed benefit vs immunoglobulins alone on disability level at 4 weeks, although one study importantly suggested possible, clinically highly relevant late effects on normalizing function. A phase 3 trial is in progress. 34 , 35 One RCT with 19 participants compared interferon beta-1a (IFNβ-1a) and placebo. It is uncertain whether IFNβ-1a improves disability after 4 weeks (very low-certainty evidence). 36
Chronic Inflammatory Demyelinating Polyneuropathy
Chronic inflammatory demyelinating polyneuropathy is an immune-mediated neuropathy that has a progressive or relapsing nature extending over 2 months. It can be divided into two types based on clinical presentation, typical and atypical. Typical CIDP presents with largely symmetrical weakness involving proximal and distal parts of extremities. Atypical CIDP takes many forms. For example, the distal acquired demyelinating symmetric (DADS) presents as symmetric length-dependent sensory or sensorimotor distal weakness with increased distal latencies; multifocal acquired demyelinating sensory and motor neuropathy [MADSAM or Lewis–Sumner syndrome (LSS)] has a multifocal distribution and the electrophysiological hallmark of conduction block, while chronic immune sensory polyradiculopathy (CISP) is restricted to sensory nerve roots only. Patients with POEMS syndrome have organomegaly, skin pigmentation, endocrinal changes, and M band in the serum. Pathophysiology of CIDP involves cellular as well as humoral immune mechanisms. Inflammatory T cells and macrophages infiltrate a nerve through the perivascular space and thus disrupt the blood–nerve barrier. 37 The humoral immune mechanism plays an equally important role; this can be inferred by the rapid response of a few CIDP patients with PE. 38 Antibodies to myelin proteins P0, P2, and PMP22, along with nodal and paranodal proteins like neurofascin NF186 and NF185, respectively, are a few of the detected target antigens in various studies. 38 , 39
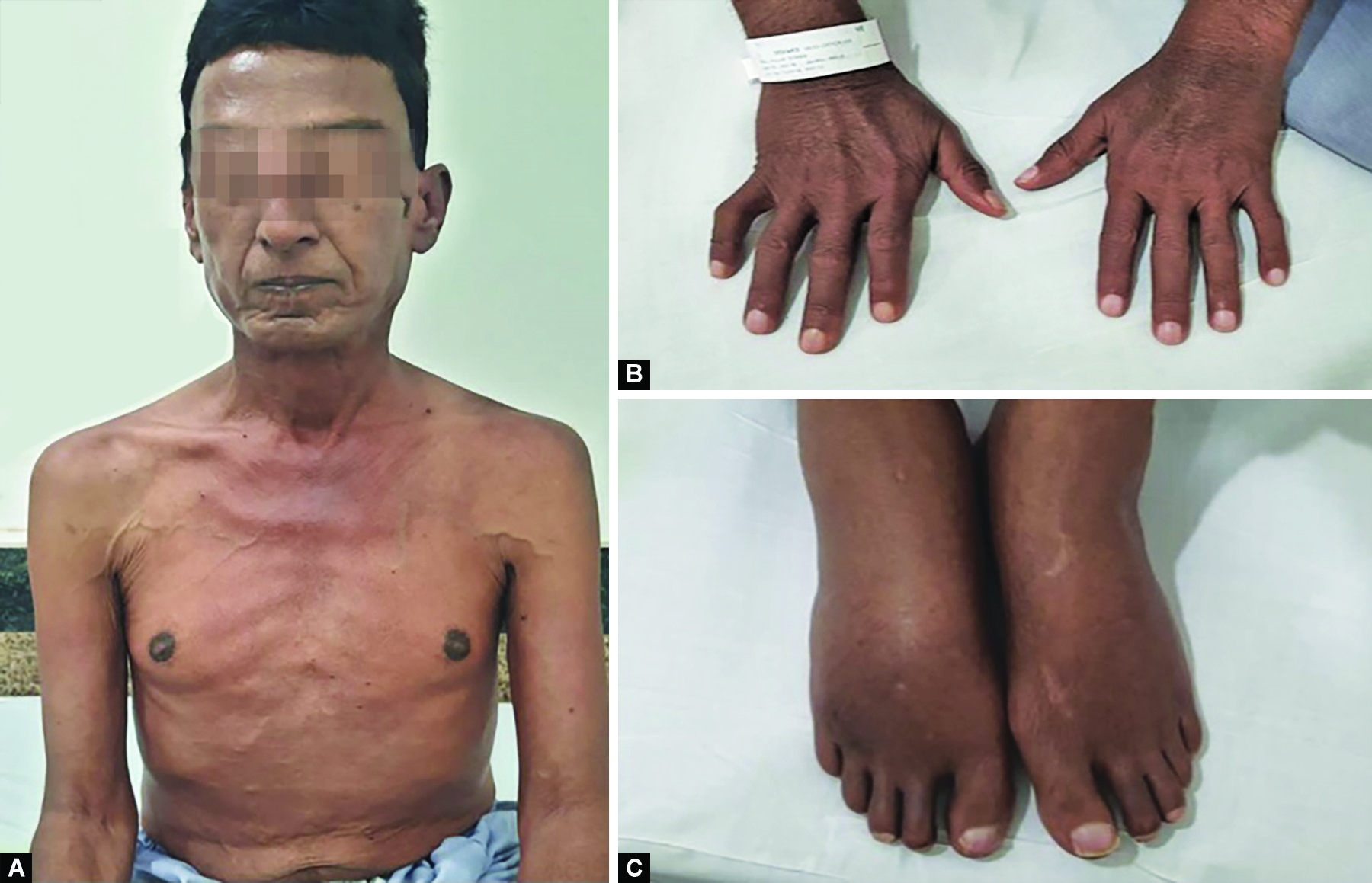
Owing to immune-mediated mechanisms, the mainstay of treatment in CIDP remains IVIG, PE, and corticosteroids. The first study to demonstrate the short- and long-term efficiency of IVIG in CIDP is the ICE study 26 (IVIG-C CIDP efficacy trial), which is a double-blind, placebo-controlled RCT of 24 weeks. The standard dose for maintenance IVIG used in this study was 1 gm/kg every 3 weeks. Subcutaneous immunoglobulin (SCIG) as a maintenance therapy has been shown to have similar efficacy as IVIG with better tolerance and convenience. This has been demonstrated in the PATH and PATH open-label extension study. 40 PE is an effective modality for CIDP as demonstrated by multiple studies, 41 , 42 but its effect lasts only for a few weeks, leading to mainly short-term benefits. IV pulse corticosteroids have been proven to show benefits in active CIDP patients. The PREDICT trial compared daily oral prednisolone with high-dose monthly dexamethasone and demonstrated moderate-quality evidence of shorter median time to improvement in the latter group. 43 Side effects like cushingoid facies, uncontrolled diabetes, and hypertension were more common in the daily oral steroid limb. IFNβ initially was considered as an adjunctive option for CIDP based on case reports, but RCT of IM IFNβ in CIDP did not show any superiority compared to the placebo. 44 Methotrexate (Mtx) as an immunomodulatory agent in CIDP was tested in an RCT recruiting 56 patients, 45 which demonstrated no benefit of Mtx as compared to a placebo. Mycophenolate mofetil (MMF) is used as an add-on steroid-sparing drug for maintenance therapy in CIDP; it is found to stabilize the clinical condition and help in reducing the steroid dose, but in a controlled trial, the agent did not show any significant improvement in modified Rankin score or muscle strength. 46 The most recent treatment modality for CIDP is rituximab (Rtx), a selective B-cell-depleting monoclonal antibody. As per a systematic review by Chaganti et al., 47 RTX was effective in 63% of CIDP patients, 48% of anti-MAG neuropathy, and 96% of patients with autoimmune nodopathy. Neurophysiological improvement was evident in 58% of CIDP and 40% of anti-MAG neuropathy patients. Rtx has been used in resistant CIDP, but it may be used earlier in the coming years based on its efficacy. Multifocal motor neuropathy, MADSAM, is treated on the lines of typical CIDP. Gammopathies need to be treated as per the primary reason for the gammopathy, and DADS patients can prove resistant to available treatment options ( Fig. 2 ).
A 55-year-old male, with atypical CIDP (distal > proximal sensorimotor weakness in all four limbs with paresthesia) along with systemic features like; (A) hepatosplenomegaly, facial hyperpigmentation, dilated veins on trunk, and fixed and raised Jugular venous pressure: (B) white nails, clubbing, and hyperpigmentation of skin; and (C) bilateral pedal edema. The patient had POEMS syndrome with monoclonal gammopathy of IgG subtype
Vasculitis Neuropathy
Vasculitis of small and medium vessels frequently affects peripheral nerves secondary to inflammation and eventually causes ischemic injury to the vasa nervorum. Vasculitis-related neuropathy can be broadly divided into systemic and nonsystemic vasculitic neuropathy (NSVN) depending on the presence of multiorgan involvement and systemic features. Peripheral neuropathy is most frequently associated with polyarteritis nodosa (PAN) and antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis. 48 Distal asymmetric, multifocal, or nonlength-dependent sensorimotor weakness, mononeuritis multiplex with lower limb predominance, is the most common pattern observed. Pain is the hallmark of vasculitic neuropathy. Nerves with moderate clinicoelectrophysiological involvement should be used for biopsy, which may show transmural inflammation with vascular damage. 49 , 50 The treatment recommendations in the Peripheral Nerve Society guidelines on NSVN are based on observational studies of NSVN and extrapolation from studies of systemic vasculitis, suggesting pulse glucocorticoids as monotherapy and the addition of other immunomodulatory agents like cyclophosphamide, Mtx, and azathioprine (AZA) for rapidly progressive NSVN. 50
INFECTION-RELATED NEUROPATHY
Neuropathy in Leprosy
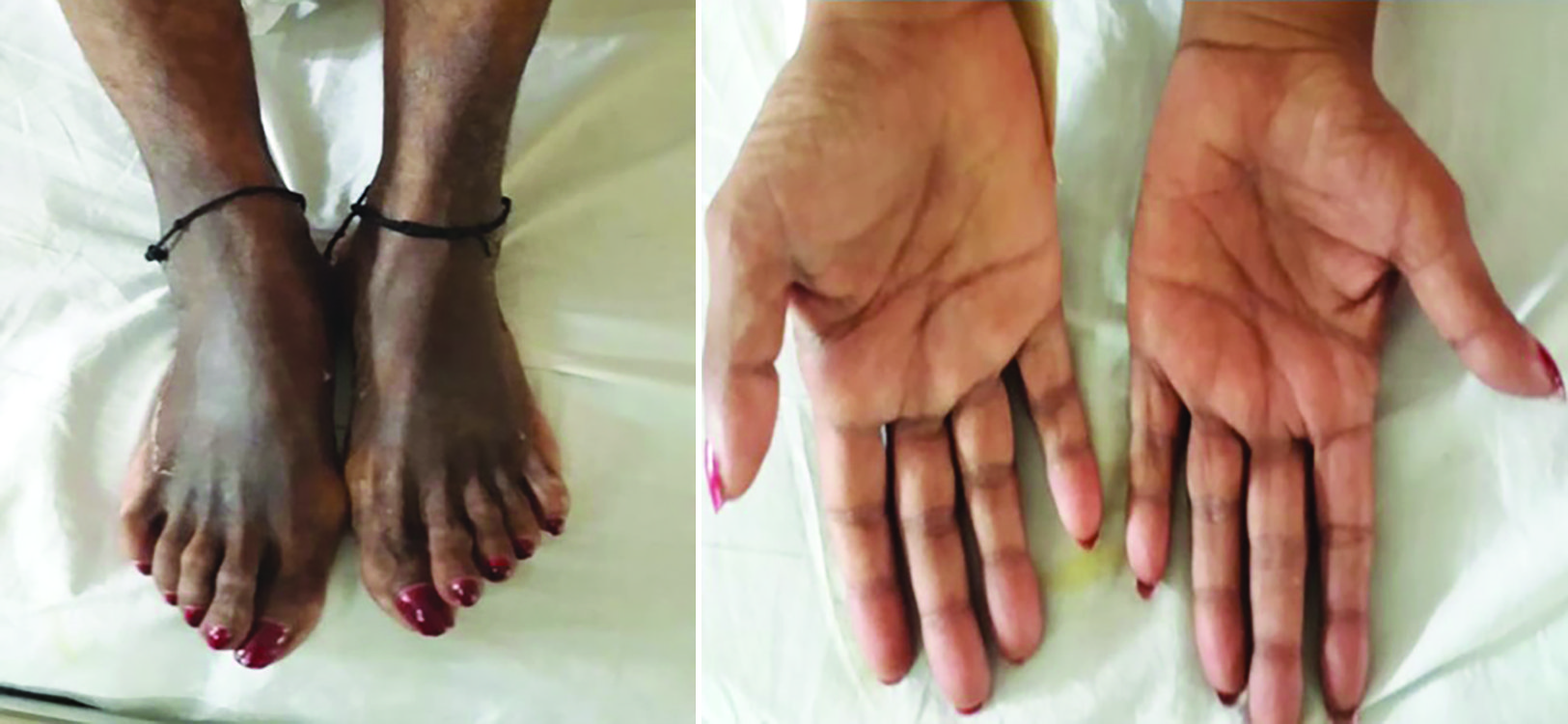
Leprosy is one of the most common treatable infectious causes of peripheral neuropathy, especially in tropical countries like India, Brazil, and other Southeast Asian countries. Leprosy classically presents with cutaneous symptoms, with a spectrum ranging from tuberculoid leprosy (TT), in which there are well-defined lesions, to lepromatous leprosy (LL) with ill-defined multiple skin lesions. Cutaneous features include hypo-/anesthetic skin patches in the buttock, back, trunk, face, and earlobes (predominantly cooler areas of the body). Pure neuritic leprosy, presenting as predominant neurologic features without significant cutaneous symptoms, causes a delay in diagnosis and treatment, thus causing increased disability related to progressive neuropathy. 51 The clinical spectrum of leprous neuropathy is wide and has been presented in Table 4 ( Fig. 3 ).
Neuropathies in leprosy
| Type of neuropathy | Clinical features |
|---|---|
| Mononeuritis and mononeuritis multiplex |
|
| Polyneuropathy | |
| Autonomic neuropathy | |
| Cranial neuropathies | |
| Acute neuritis |
|
| Ganglionitis |
|
A 40-year-old female with skin changes in LL secondary to autonomic neuropathy (A) with mononeuritis multiplex—wasting of left hand, and (B) Photo courtesy—Dr Varsha Patil
Diagnosis of leprosy is mostly clinical, but tests like nerve conduction, skin and nerve biopsy, nerve ultrasonography, and MR neurography help confirm the clinical findings. The WHO Expert Committee on Leprosy defines a case of leprosy as an individual who has one of the following cardinal signs of leprosy but who has not received a full course of multidrug therapy (MDT) appropriate for the type of leprosy. 60
Definite loss of sensation in a pale (hypopigmented) or reddish skin patch.
A thickened or enlarged peripheral nerve with a loss of sensation and/or weakness in the muscles supplied by the nerve.
The presence of AFB in slit skin smears.
Treatment
Medical treatment of leprosy.
Management of neuropathic pains.
Medical Management of Leprosy
As per WHO recommendations, patients fulfilling the diagnostic criteria should be started on MDT including three drugs (dapsone, rifampicin, and clofazimine) for all leprosy patients irrespective of bacillary load. The duration for paucibacillary leprosy is 6 months, whereas for multibacillary it is 12 months.
Table 5 depicts the treatment regimens for various types of leprosy.
Treatment schedules in leprosy 60
| Paucibacillary | Multibacillary | |
|---|---|---|
| Drugs | Dapsone 100 mg daily and clofazimine 50 mg daily Rifampicin 600 mg monthly and clofazimine 300 mg monthly | Dapsone 100 mg and clofazimine 50 mg daily Rifampicin 600 mg and clofazimine 300 mg monthly |
| Duration | 6 months | 12 months |
Drug-resistant Leprosy
Although not very common, drug-resistant leprosy has been documented. WHO recommends the use of second-line medications such as ofloxacin, minocycline, clofazimine, and clarithromycin. 61
Usually, leprosy presents with predominantly negative symptoms like numbness or anhidrosis, but during typical lepra reactions, neuropathic pains can be particularly predominant. Corticosteroids are the definitive treatment to reduce the impact of nerve damage, and they also help alleviate the neuropathic pains. Symptomatic treatments that can be offered are gabapentin, pregabalin, and duloxetine. Lepra reactions are treated with high doses of corticosteroids over a few weeks to months, and in resistant cases, clofazimine and thalidomide can be used. 51
Human Immunodeficiency Virus-related Neuropathy
Among other neurological features of HIV1, peripheral neuropathy is the most common neurological complication, affecting 30–50% of individuals. 62 , 63 HIV-associated neuropathies may present with varied clinical presentations at different stages of illness. DSPN is the most common, but inflammatory demyelinating neuropathies, progressive polyradiculopathies, mononeuritis multiplex, autonomic neuropathy, and the highly active antiretroviral therapy (HAART)-related neuropathies are encountered from time to time.
Treatment of Human Immunodeficiency Virus-related Neuropathy
Human immunodeficiency virus-related neuropathy can be prevented with adequate suppression of viral load and an increasing trend of CD4 count. The evolution of sensory neuropathy after HAART was observed by Centner et al. in 2017. 64 Results indicated that painful symptoms improved after long-term neuro-safe HAART via reduction of exposure to HIV-induced oxidative stress. HAART-induced neuropathy, commonly reported with stavudine, didanosine, nevirapine, zalcitabine, and protease inhibitors, requires switching over to newer drugs for effective management of HIV as well as to help prevent the risk of neuropathy. Lamivudine, abacavir, dolutegravir, emtricitabine, and adjusted doses of protease inhibitors are safer in this regard. Neuropathic pain is the most disabling complaint in HIV-related neuropathy, and drugs like antidepressants (tricyclics), anticonvulsants (gabapentin, pregabalin, lamotrigine), and topical analgesics have been successfully used in the treatment of neuropathic pain. HIV-related DSP has a better response from capsaicin 8% cutaneous patch when compared to mononeuropathy, cervical radiculopathy, and postherpetic neuralgia. 65 Other nonpharmacological interventions like hypnosis, 66 dietary supplements like curcumin and tart cherry extracts, 67 and bromelain 68 which increase the antioxidants and reduce neuronal stress are still under investigation but can be used in refractory conditions with caution.
DRUGS/TOXIN-INDUCED NEUROPATHY
Toxic peripheral neuropathies can occur secondarily to environmental, occupational, recreational, and iatrogenic (drug-induced) causes. The most common pattern observed is distal symmetric length-dependent sensory neuropathy along with motor or autonomic neuropathy. Several prescription medicines are known to cause neurotoxicity, like chemotherapy agents and antibiotics such as isoniazid, metronidazole, and nitrofurantoin. Alternative medicine products have been shown to contain heavy metals like lead, mercury, and arsenic, which are known to cause neurotoxicity along with other systemic features. Table 6 presents the important aspects of common drug-induced neuropathies.
Important aspects of common drug-induced neuropathies
| Drugs | Incidence | Site of toxicity | Clinical features | Treatment |
|---|---|---|---|---|
| Chemotherapy drugs | ||||
| Cisplatin | 30% of symptomatic neuropathy | Sodium channel abnormalities causing axonal hyperexcitability and repetitive discharges ± DRG | First symptoms appear after 1st month of treatment Dose-dependent toxicity Chronic sensory predominant small fiber neuropathy with burning paresthesia, pain, and tingling | After stopping the drug, autonomic and motor symptoms tend to improve. Sensory symptoms worsen after finishing the chemotherapy—coasting stage. Eventually coming back to baseline 69 |
| Oxaliplatin | 10–20% with modest dose | Sodium channel abnormalities | Neurotoxicity presents as two types—acute and chronic sensory neuropathy Cold-induced sensitivities | Acute neuropathy subsides after stopping the drug, and increases after consecutive dose—Sawtooth pattern of neuropathy 70 |
| Vincristine | Almost all | Microtubular axon transport function abnormalities DRG | Sensory predominant with mild motor weakness (weakness in finger extension first) 71 Sensory ataxia ± 72 | Symptomatic treatment for small fiber neuropathy like gabapentin, pregabalin, and lamotrigine |
| Bortezomib | 30–60% | DRG Small fibers, type C | Along with small fiber neuropathy, bortezomib also presents with severe motor-predominant polyradiculopathy 73 | Symptomatic treatment |
| Antibiotics | ||||
| Metronidazole | 5–6% | Nerve | Sensory predominant reversible length-dependent neuropathy Predominantly negative symptoms—numbness and decreased sensations | Stopping the drug usually reverses neuropathy 74 |
| Isoniazid | 10–20% | Nerve | Starts with tingling numbness and decreased sensation distally followed by a glove and stocking-like pattern | Prevented with pyridoxine prophylaxis. Reverses if stopped with early symptoms 75 , 76 |
| Dapsone | 1–2% | Motor | Upper limb predominant multiple mononeuropathies 77 | Stop the drug |
| Nitrofurantoin | Incidence is higher in the elderly and patients with renal disease | Sensory nerve Demyelinating | GBS like syndrome Length-dependent sensorimotor polyneuropathy 78 , 79 Sensory painful DSPN | Most neuropathy related to nitrofurantoin are refractory and have delayed improvement after stopping the drug |
| Linezolid | As high as 80% | Sensory painful small fiber neuropathy | Dose-dependent toxicity. Small fiber burning, pain, and paraesthesia 78 , 80 , 81 | The improvement is mostly seen after 6 months of cessation of the drug. Few can be irreversible |
| Heavy metals | ||||
| Lead | 30% | Axonal damage of motor nerves | Acute to subacute form—predominantly motor neuropathy—frequently starts with distal extension weakness in upper limbs Chronic long-term exposure—mild sensory and autonomic neuropathy | Acute neuropathy—chelation is helpful to some extent. Neurotoxic features are not reversible in chronic forms 82 , 83 |
| Arsenic | GBS like syndrome | Acute form—GBS-like syndrome, although cranial nerve involvement is rare Chronic form—length-dependent sensory motor axonal neuropathy | Chelation may help with the acute form of neuropathy. Treatment of GBS-like syndrome involves PE or IVIG | |
| Mercury | Peripheral nerve axons CNS | Sensory predominant and ataxia Behavioral changes | Chelation and cessation of exposure | |
| Alcohol | Peripheral nerve | Malnutrition—involves multiple vitamin deficiencies like thiamine and Vit B12, causing sensorimotor peripheral neuropathy Without malnutrition—mild form of distal sensory polyneuropathy | Abstinence improves early symptoms | |
| Pyridoxine excess | DRG/axons | Sensory axonopathy or neuronopathy | ||
| Organophosphorus compounds | Motor > sensory axons | Distal motor weakness, foot drop, cramps, and mild sensory disturbances | Symptomatic management |
CRANIAL NEUROPATHIES
Various disease categories such as infections, inflammations, tumors, and infiltrations affect the cranial nerves. The commonly encountered conditions are as follows. Among the infections, the herpes zoster virus affects the facial nerve, causing Bell's palsy, and the seventh and eighth nerves, causing Ramsay Hunt syndrome. In children, diphtheria can result in lower cranial neuropathies, producing dysphagia. Tuberculosis of the nervous system, mainly meningitis, is common, and the basal exudates result in various cranial neuropathies, often predominating in the lower segments. Common inflammations are the Tolosa Hunt syndrome, which results from inflammation at the apex of the orbit. A proportion of such patients have the IgG4 antibodies. Patients respond to corticosteroids but may have a recurrent and prolonged course requiring long-term immunotherapies. Lymphomas, leukemias, other lymphoreticular malignancies, and deposits from other bodily tumors can infiltrate or compress the cranial nerves, resulting in their dysfunction. Cerebrospinal fluid centrifuge examination for abnormal cells and contrast MRI scans help the diagnosis, and the treatment depends upon the primary condition. The optic nerve, being a part of the central nervous system myelin, is affected by demyelinating diseases such as multiple sclerosis and neuromyelitis optica, and toxic and deficiency diseases such as the B12 deficiency. Toxic amblyopia and methanol toxicity are some common examples in clinical practice.
ENTRAPMENT NEUROPATHIES
Entrapment neuropathies are treatable disorders that are caused by compression of peripheral nerves secondary to passage through narrow anatomical spaces. They are characterized by pain and/or sensorimotor loss. The common pathomechanisms of entrapment neuropathies are as follows 84 :
Extra- and intraneural ischemia.
Demyelination and axonal degeneration.
Neuroinflammation.
These compression neuropathies can be divided based on gradings of peripheral nerve injuries such as neuropraxia (nerve sheath intact but nerve function is temporarily impaired), axonotmesis (nerve fibers are interrupted but connective tissue remains intact), and neurotmesis (most severe type of nerve injury where the nerve fibers and connective tissue are severed). A few of the common entrapment neuropathies are described in Table 7 .
Entrapment neuropathies of upper and lower limbs
| Nerve entrapped | Anatomical site of compression | Clinical presentation | Treatment |
|---|---|---|---|
| Upper limbs | |||
| Long thoracic nerve (C5–C7) |
| Medial winging of the scapula | Thoracic/supraclavicular decompression or combined—within 6–12 months for better results 85 |
| Spinal accessory nerve (C1–C6) | At the jugular foramen—due to tumor In the posterior triangle—due to lymph nodes/intervention | Lateral winging of scapula | Medical management for pain Surgical decompression—resection of fascia 86 |
| Axillary nerve (C5–C6) | Quadrilateral space by posterior humeral circumflex artery | Paresthesias, posterior shoulder pain, weakness of deltoid, and teres minor | Medical management for pain and paresthesias Surgical intervention within 6–12 months of onset 87 |
| Median nerve (C6–T1) | AIN entrapment—pronator teres heads Carpel tunnel syndrome—flexor retinaculum at wrist | AIN—weakness of muscles in deep anterior compartment CTS—dull aching pain at wrist, worsened in night, with paresthesias in later 3.5 fingers | Severe sensorimotor weakness—surgical intervention such as decompression of flexor retinaculum relieves maximum symptoms Mild-moderate weakness—medical management with gabapentin, pregabalin, etc. 88 |
| Ulnar nerve (C8–T1) | Medial epicondyle and olecranon—cubital tunnel syndrome Guyon canal syndrome—palmar carpel ligament | Pain in medial elbow with intermittent numbness in ring finger and little finger with weakness of small muscles of hands | In situ decompression, medial epicondylectomy, anterior subcutaneous transposition, intramuscular transposition, and submuscular transposition 89 |
| Radial nerve (C5–T1) | Arcade of Frohse Deep head of supinator muscle | Lateral elbow pain and wrist drop | Decompression surgery—relieving the nerve from arcade of Frohse—good prognosis 90 |
| Lower limbs | |||
| Sciatic nerve (L4–S3) |
| Posterior thigh and hip pain, ± radicular in nature. Buttock pain aggravated by sitting—compression by piriformis muscle | Most cases—resolve spontaneously Bed rest Analgesics/NSAIDs, physical and behavioral therapy Interventions like chemonucleolysis for refractory leg pain may be tried 91 |
| Lateral femoral cutaneous nerve of thigh (L1–L3) | Most common compression at exit from pelvis | Unilateral pain, paresthesia, and numbness in the lateral or anterolateral thigh, relieved after sitting 92 | Conservative treatment focuses on reduction of the factors that cause or intensify nerve compression, for example avoidance of tight clothing and constrictive belts around the waist Surgical intervention-decompression of nerve at inguinal ligament 92 |
| Peroneal nerve |
| Foot drop, pain, and numbness of the lateral lower leg and foot dorsum, aggravated by plantar flexion and foot inversion | Timely surgical decompression can treat peroneal entrapment neuropathy, positive correlation between decreased time of surgery, and better outcomes 93 |
AUTONOMIC NEUROPATHY
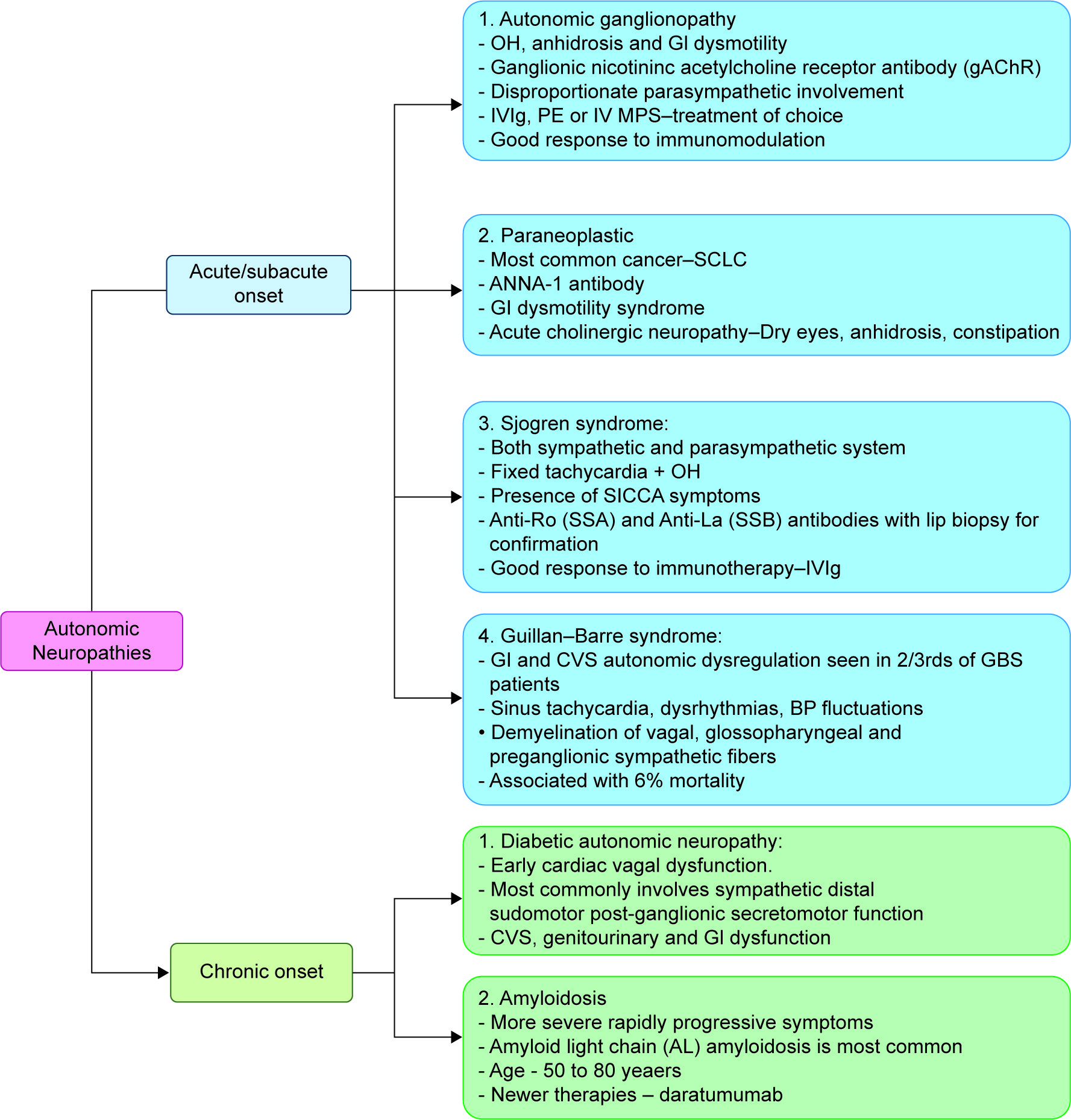
Autonomic neuropathies are a complex group of disorders targeting mainly the autonomic fibers, including the parasympathetic and sympathetic systems. They are classified according to temporal course/disease onset (acute/subacute/chronic), etiology (hereditary/acquired), and as per the involvement of the fibers (parasympathetic/sympathetic/generalized). 94 Pertaining to the current article, a few of the most important treatable entities are described briefly in Figure 4 .
Autonomic neuropathies
CONCLUSION
As can be surmised from the above discussion, a variety of neuropathic processes have the potential of reversibility, and these need to be rapidly identified and treated for best outcomes. Evaluation of clinical features, coupled with investigations, helps the process of segregation and reaching the diagnosis. Inflammatory, infective, toxic, metabolic, and deficiency neuropathies form the main categories of reversible neuropathies and should be actively looked for for best outcomes.
ACKNOWLEDGMENT
Photo courtesy to Dr Varsha Patil—Consultant Neurologist at Bombay Hospital Institute of Medical Sciences, Mumbai.
AUTHOR CONTRIBUTIONS
SVK: concept, design, and review.
JM: literature review and writing the manuscript.
HH: design, manuscript preparation, and review.
RR: concept and review.
REFERENCES
ARTICLE INFO
Publication history
received: 19 February, 2025
accepted: 08 May, 2025
Published: 01 September, 2025
Copyright
Copyright © 2025; The Author(s).